The Effect of Arteriovenous Dialysis Access Creation on Pulmonary Artery Pressures in Hemodialysis Patients
Kalyan Janga1, Sheldon Greenberg1, Elie Fein1, Ilya Shneyderman1, Gerald Hollander2, Anil Hingorani3, Winston Lee1
1- Division of Nephrology, Maimonides Medical Center. Brooklyn, NY
2- Division of Cardiology, Maimonides Medical Center. Brooklyn, NY
3- Division of Vascular Surgery, Maimonides Medical Center. Brooklyn, NY
Corresponding Author: Winston Lee
2651 East 14th Street
Brooklyn, NY 11235
Abstract
Background:
Pulmonary hypertension (PH) is a significant cause of cardiovascular morbidity and mortality. PH in the setting of end stage renal disease (ESRD) is a phenomenon which recently has been the subject of investigation. The presence of high flow AVF has been linked with PH. We attempt to further elucidate the relationship between AVF and progression of PH in ESRD patients; specifically the elevation of pulmonary systolic pressure after the creation of a hemodialysis access.
Methods:
We reviewed the vascular surgery database of a tertiary care medical center. We identified individuals who had a hemodialysis access placed from March 1994 to April 2007. These records were compared with the echocardiogram database. Individuals who had two echocardiograms performed at least 3 months prior to and 3 months following the access placement were analyzed. Pulmonary pressures prior to and post dialysis access placement were compared.
Results:
61 subjects were identified for analysis. 28 out of 61 (45.9%) ESRD patients had worsening of pulmonary artery pressures. In evaluating incident pulmonary hypertension, the ESRD group had 16/61 (26.2%). Tthe presence of an avf vs avg showed no change in progression of PH: 21/46 (45.7%) with AVF vs 7/15 (46.7%) with AVG. Location of dialysis access was associated with progression of PH. 23/42 subjects had worsening pulmonary pressure with upper arm access versus 4/16 for lower arm access (p=0.014).
Conclusions:
According to our data, there may be a correlation between placement of dialysis access and increasing of pulmonary artery pressures.
Key Words:
Dialysis; pulmonary hypertension; AV fistula
Introduction
Pulmonary hypertension (PH), which has been defined as pulmonary artery pressure greater than 25mm of Hg at rest and 30mm of Hg during exercise[1], is a significant cause of cardiovascular morbidity and mortality. PH in the setting of end stage renal disease (ESRD) and hemodialysis is a phenomenon which recently has been the subject of investigation. It has been reported that the prevalence of PH in patients receiving hemodialysis may be as high as 40%-51%[2,3]. Arteriovenous fistulae (AVF) are the preferred access for those undergoing hemodialysis. The presence of high flow AVF has been linked with PH; the link in ESRD patients has also been previously described [2]. The etiology of elevated pulmonary pressures in this population is likely multifactorial. The presence of renal disease on its own may predispose to PH; prevalence of PH predialysis patients has been found to be 39.1%[4] and in peritoneal dialysis patients has been estimated to be anywhere from 12.6% to 42%[5,6]. We attempt to further elucidate the relationship between AVF and development and progression of PH in ESRD patients; specifically the elevation of pulmonary systolic pressure after the creation of a hemodialysis access.
Subjects and methods
We performed a retrospective cohort study utilizing the vascular surgery database of a single, tertiary care medical center. We identified individuals who had a hemodialysis access placed from March 1994 to April 2007. These records were then cross referenced with the echocardiogram database of that medical center. Individuals who had two echocardiograms performed at least 3 months prior to and 3 months following the access placement were analyzed.
Pulmonary artery pressures were estimated noninvasively by calculating the transtricuspid gradient, right atrial pressure, inferior vena cava diameter and inferior vena cava inspiratory collapse. Pulmonary hypertension classifications were as follows: mild (36- 50 mm Hg), moderate (51-70 mm Hg) and severe (>70 mm Hg). Final cardiologist readings further classified pulmonary hypertension as mild-moderate and moderate-severe (pulmonary pressure equivalents not given). Pulmonary pressures prior to and post dialysis access placement were compared.
General demographic data (age, sex, race), type of access (fistula vs graft) and location of access (upper vs. lower arm) were also used as variables in the analysis. Patients with significant preexisting valvular disease or ventricular dysfunction (coded as more than mild in the echocardiogram report) were excluded. Data was analyzed via chi squared with SPSS software v 14. The study was performed in accordance with the Declaration of Helsinki. Informed consent was waived for this retrospective chart review.
Results
Initially 119 individuals with newly placed dialysis access and multiple echocardiograms were identified. Of these, 58 were excluded for baseline ventricular dysfunction or valvular disease. Median time between echocardiograms was 17.6 months. Median time from access surgery to repeat echocardiogram was 11 months. Of ESRD patients, 46 (74%) had avf placed, while 15 (26%) had arteriovenous grafts (avg). Of the 58 ESRD patients with data available on access position, 42 had forearm accesses (72%) and 16 upper arm accesses (28%).
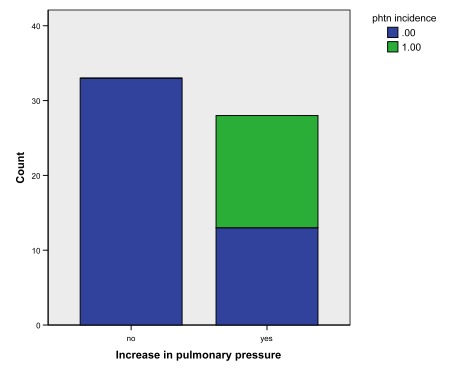
Results are seen in figure 1. When using an increase of one whole category (e.g. mild to moderate) to define progression of pulmonary hypertension, 28 out of 61 (45.9%) ESRD patients progressed. In evaluating new onset of pulmonary hypertension during the time frame; the ESRD group had 16/61 (26.2%). Further subgroup analysis shows that the presence of an avf vs avg showed no change in progression of PH: 21/46 (45.7%) with AVF vs 7/15 (46.7%) with AVG. Location of dialysis access was significantly associated with progression of pulmonary hypertension. 23/42 subjects had worsening pulmonary pressure with upper arm access versus 4/16 for lower arm access (p=0.014).
Prior to applying exclusion criteria, overall prevalence of PH at time of second evaluation was 56.8%. In patients without significant ventricular dysfunction or valvular disease, the prevalence of PH was 52.5%.
Figure 1. Number of subjects with increased pulmonary pressures
Green portion represents those subjects who had no evidence of pulmonary hypertension at first echo.
Discussion
Pulmonary hypertension remains a significant cause of morbidity and mortality in the ESRD population, and the natural history of this condition remains poorly understood in this group. As mentioned previously, there are several factors that may influence the worsening of pulmonary artery pressures in dialysis patients. There is some evidence of decreased nitric oxide production in hemodialysis patients with arteriovenous fistulas (AVF) and PH, which may contribute to increased tone in the pulmonary vasculature [7]. Elevated levels of other inflammatory cytokines such as IL-1 beta, TNF-alpha and IL-6 have also been found in this population [8]. The hemodynamic effect of these AVF has been well documented. Clinical and hemodynamic improvement has been shown when fistulae (dialysis related or otherwise) have been closed (either temporarily or permanently)[2, 9-11].
In this study we attempted to determine the effect placing a dialysis arteriovenous access on pulmonary arterial pressure. Slightly less than half (28/61-45.9%) of hemodialysis patients without significant underlying ventricular dysfunction or valvular disease had an increase in their pulmonary artery pressures, as measured by echocardiogram. Although there was no matched control group for these subjects, we did select a group of patients from the echocardiogram database who had two echocardiograms in the same time frame. The increase in pulmonary pressures in the dialysis group was significantly more than the non dialysis group. When selecting the cutoff as an increase of a half category (i.e. mild to mild-moderate), 33/61 (54.1%) of ESRD patients had an increase in pulmonary pressures. There was also a trend towards increasing incident cases of pulmonary hypertension in the ESRD group. Upon further analysis presence of upper arm access was significantly associated with increase in pulmonary pressures as compared to lower arm access. Physiologically, this may be due to higher flows through the access, and higher cardiac output. Although this relationship would seem to explain this finding, there has been a small study of 32 ESRD patients that did not show a correlation between AVF flow and pulmonary artery pressures [12].
Our results are similar to those of Yigla, et al. [13] who performed a small prospective study of 12 patients looking at echocardiography before and after access surgery. Their prevalence of PH was 40%, and 40% of the patients (5/12) had worsening of their pulmonary pressure of at least 10mm Hg after the fistula placement.
There are several important limitations to our study; the fact that it is a retrospective cohort analysis made it difficult to capture several variables that would be of interest. Specifically, presence of chronic obstructive pulmonary disease (and other underlying pulmonary diseases) which may confound the results significantly. It would have also been informative to have data on the blood flow through the dialysis access to determine possible correlation with pulmonary pressures. The retrospective nature also limited us in terms of more specific data on pulmonary pressures, as well as identifying equivalent control group for the dialysis patients. We also acknowledge that these patients are specifically selected for multiple echocardiograms; this may lead to an overestimation of progression of PH compared to the general dialysis population.
AVF are the preferred conduits for hemodialysis because of improved clearance, longevity and decreased morbidity compared to grafts and catheters. However, pulmonary hypertension is an increasingly recognized complication in ESRD patients. A study of 127 dialysis patients found that development of PH both before and after initiation of hemodialysis was an independent risk factor for mortality[14]. According to our data, there may be a correlation between placement of dialysis access and increasing of pulmonary artery pressures. While it is unrealistic and likely counterproductive to limit widespread use placement of AVF, certain specific cases may warrant further investigation. In those who have underlying lung disease or preexisting PH it may be reasonable to stress lower arm fistulae, or consider alternative forms of therapy, such as peritoneal dialysis. Further prospective studies outlining the specific impact on morbidity and mortality are clearly needed in this area.
References
1. Galie N, Torbicki A, Barst R, et al. Guidelines on the diagnosis and treatment of pulmonary arterial hypertension. The Task Force on Daignosis and Treatment of Pulmonary Arterial Hypertension of the European Society of Cardiology. Eur Heart J. 2004;25:223-2278
2. Yigla M, Nakhoul F, Sabag A, Tov N, Gorevich B, Abassi Z, Resiner S. Pulmonary hypertension in patients with end-stage renal disease. Chest. 2003 May;123(5):1577-82
3. Mahdavi-Mazdeh M, Alijavad-Mousavi S, Yaytazadeh H, Azadi M, Yoosefnejad H, Ataiipoor Y. Pulmonary hypertension in hemodialysis patients. Saudi J Kidney Dis Transpl. 2008 Mar;19(2):189-93
4. Havlucu Y, Kursat S, Ekmekci C, Celik P, SerterS, Bayturan O, Dinc G. Pulmonary hypertension in patients with chronic renal failure. Respiration. 2007;74(5):503-10
5. Yasuda T, Tanabe N, Konishi K, Shigeta A, Shinohara M, Toyama S, Nakamura M, Maruoka M, Tada Y, Takiguchi Y, Tatsumi K, Kuriyama T. [Pulmonary hypertension in a patient on chronic hemodialysis] (abstract). Nihon Kokyuki Gakkai Zasshi. 2009 Jan;47(1):52-6
6. Kumbar L, Fein PA, Rafiq MA, Borawski C, Chattopadhyay J, Avram MM. Adv Perit Dial. 2007;23:127-31
7. Nakhoul F, Yigla M, Gilman R, Reisner SA, Abassi Z. The pathogenesis of pulmonary hypertension in haemodialysis patients via arterio-venous access. Nephrol Dial Transplant. 2005 Aug;20(8):1686-92
8. Yu TM, Chen YH, Hsu JY, Sun CS, Chuang YW, Chen CH, Wu MJ, Cheng CH, Shu KH. Systemic inflammation is associated with pulmonary hypertension in patients undergoing hemodialysis. Nephrol Dial Transplant. 2009 Jun;24(6):1946-51
9. Clarkson MR, Giblin L, Brown A, Little D, Donohoe J. Reversal of pulmonary hypertension after ligation of a brachiocephalic arteriovenous fistula. Am J Kidney Dis. Sep;40(3):E8
10. Kolilekas L, Gallis P, Liasis N, Anagnostopoulos GK, Eleftheriadis I. Unusual case of pulmonary hypertension. Respiration. 2006;73:117-19
11. Bhatia S, Morrison JF, Bower T, Mcgoon MD. Pulmonary hypertension in the setting of acquired systemic arteriovenous fistulas. Mayo Clinic Proceedings. 2003;78:908-12
12. Acarturk G, Albayrak R, Melek M, Yuksel S, Uslan I, Atli H, Colbay M, Unlu M, Fidan F, Asci Z, Cander S, Karaman O, Acar M. The relationship between arteriovenous fistula blood flow rate and pulmonary artery pressure in hemodialysis patients. Int Urol Nephrol. 2008;40(2):509-13
13. Yigal M, Banderski R, Azzam ZS, Reisner SA, Nakhoul F. Arterio-venous access in end-stage renal disease patients and pulmonary hypertension. Ther Adv Resp Dis. 2008;2(2):49-53
14. Yigal M, Fruchter O, Aharonson D, Yanay N, Resiner SA, Lewin M, Nakhoul F. Pulmonary hypertension is an independent predictor of mortality in hemodialysis patients. Kidney Int. 2009 May;75(9):969-75
Copyright Priory Lodge Education Limited 2010
First Published July 2010
All pages copyright ©Priory Lodge Education Ltd 1994-


